'We took it to the real world and it worked': Rapid Zika testing platform clears hurdle with Brazil trial
Published: March 16, 2022
An international team of researchers, led by experts from the University of Toronto’s Leslie Dan Faculty of Pharmacy, has conducted one of the first field trials for a synthetic biology-based diagnostic platform that could provide rapid, de-centralized and low-cost patient testing for infectious diseases such as the Zika virus.
The work, conducted on-site in Latin America, revealed the potential for the cell-free synthetic biology tools and companion hardware. Published in Nature Biomedical Engineering, the study’s results show that the novel diagnostic platform has analytical specificity and sensitivity equivalent to a U.S. Centres for Disease Control (CDC) PCR test for Zika and a diagnostic accuracy of 98.5 per cent with 268 patient samples collected in Recife, Brazil.
The platform is also programmable and can be similarly applied to detect any pathogen sequence. In addition to validating highly accurate diagnostic results for Zika, the team also achieved similar diagnostic performance for chikungunya virus, another mosquito-borne arbovirus.
“We see emerging diagnostics, like the paper-based tests we’ve developed, as having tremendous near-term potential to augment existing PCR capacity, improve equity in access to health care and aid in the responses to public health crises,” said Keith Pardee, assistant professor in the department of pharmaceutical sciences in the Leslie Dan Faculty of Pharmacy.
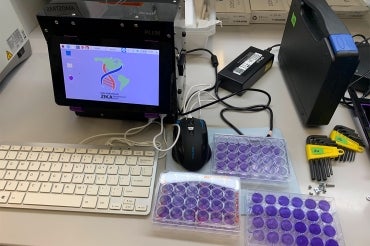
The portable diagnostic platform is a combination of a cell-free, paper-based test and a field-ready companion device that allows data to be collected using image-based colour analysis – purple for positive and yellow for negative. Called “PLUM” (Portable, Low-cost, User-friendly, Multimode), the toaster-size reader presents results from up to 384 samples and displays them in a single image capture.
Prior to the current global COVID-19 pandemic, the 2015-2016 outbreak of Zika virus in Latin America emphasized the urgent need for rapid and low-cost testing that can be deployed beyond the reach of centralized diagnostic labs, according to Pardee, a Canada Research Chair in Synthetic Biology and Human Health.
“We were investigating and developing this technology well before the COVID-19 pandemic brought these issues to light at the global level,” he said. “We’ve now been able to apply it and validate it in a region of endemic disease, which is really promising because these tools are meant to enable health systems to better respond to future outbreaks of infectious disease, particularly in low-resource settings.”

From left: Margot Karlikow, Seray Çiçek, Livia Guo and Keith Pardee (photo courtesy of Livia Guo, LSK Technologies)
Lindomar Pena, of the department of virology at the Oswaldo Cruz Foundation (Fiocruz) in Rio de Janeiro, led the Brazilian team that collaborated on the project.
“This robust diagnostic platform displayed desirable features to be used in developing countries such as Brazil and in laboratories with basic infrastructure,” Pena said. “We hope it can be further developed and deployed in the Brazilian network of public health laboratories to diagnose Zika patients, trace contacts and identify hot-spot areas with active community transmission.”
The hardware and software that make up PLUM were originally developed by co-authors Livia Guo and Seray Çiçek as part of their graduate work in the Pardee lab. To keep production costs low, Guo and Çiçek – who are co-founders of LSK Technologies, a startup that aims to commercialize PLUM – used customizable software programs and off-the shelf electronics, enabling the device to be built for approximately US$500 per unit.
On the molecular side, the cell-free tests can be freeze-dried, allowing for distribution without refrigeration and, significantly, all of the molecular components of the test are independent of the PCR-supply chain.
“Here we have demonstrated that these two technologies combined create a low-cost, highly accurate diagnostic tool,” said study lead author Margot Karlikow, a post-doctoral researcher in the Pardee lab from 2016 to 2021 and now co-founder of En Carta Diagnostics.
“We also demonstrated that it is feasible to transport the platform across a significant distance and implement it effectively in another country. In many low- and middle-income countries, there is no PCR testing available outside of main cities, so the ultimate goal is that this platform be used as a high-quality alternative to PCR in more regional settings.”
As part of their publication, the team shared a roadmap of how they were able to transport the diagnostic technology from Canada to Latin America so that other teams might benefit from their experiences.
Guo, Çiçek and Karlikow successfully moved their work with the Pardee lab to establish health tech startups, LSK Technologies and En Carta Diagnostics, respectively. The entrepreneurship and incubator community at U of T was instrumental in the process.
In particular, Guo and Çiçek collaborated closely with the H2i and UTEST incubators.
“Both H2i and UTEST were great in terms of making introductions and helping to expand our network,” said Çiçek, explaining that H2i helped the team figure out a road map from the regulatory and product design standpoint while UTEST helped with customer discovery so they could better understand and mitigate customer pain points.
“The entrepreneurship community at U of T helps foster regular and valuable discussions between entrepreneurs around their challenges,” said Karlikow who is now running En Carta Diagnostics from Paris. “With so much support available, it helps you to make the decision to start your company to push your vision and product forward.”
Showing that the platform could be transported and accurately detect Zika virus in patient samples is a significant step forward in creating more accessible and de-centralized testing, says Pardee.
However, the extraction of RNA from patient samples still requires liquid handling by skilled technicians at this stage. “With performance on patient samples now validated, we are tackling these next challenges, like sample preparation, so that the platform and PCR-like diagnostic capacity can be distributed more broadly into the communities where they are needed.”
The team is hoping the successful field-based patient trial will move the work forward with added momentum.
“This step of translating the technology from the lab and applying it in a real-world setting was so important,” said Karlikow. “This was more than just the science involved – we made it applicable. We took it to the real world and it worked.”



